where x is the time the temperature was taken and y is the temperature.
We used trial and error to construct this function, focusing on decreasing
the measure of error between the model and the observed data. This measure
of error was found by taking the square of the difference for each time,
summing the squares, and dividing by the number of data points (i.e., the
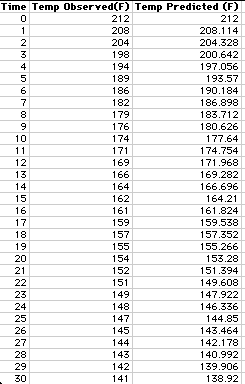
variance). The measure of error was decreased to 7.082. The following table
shows the observed and predicted temperatures.
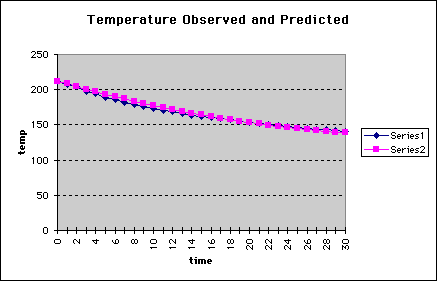
The following graph shows the observed data (Series 1-blue) and the predicted
data (Series 2-pink).
Although the measure of error between the model and the observed data
is 7.082, we get the following predicted temperatures after 45, 60 and 300
minutes respectively: 136.12, 155.84, and 3531.2. If we look at the function
used to predict the data, we see that the time is squared. Thus at time
= 300, we would expect the predicted temperature to be HUGE!!!!!!!! SO THIS
IS A BAD MODEL to use for predicting later temperatures, yet it is quite
good within our time range!!!!
In conclusion, there are many factors in this investigation that can have
an effect on the data. For instance, the type of container used to hold
the water, the temperature of the room, the type of thermometer used (e.g.,
the scale on the thermometer could be different from the one used in this
investigation), and human error in reading the thermometer all can have
some effect on the data collected. Thus, given different conditions as well
as different data collectors, we could obtain different results.
For application to the classroom, we could have students perform this investigation
at home and then compare data and make inferences from their findings. For
example, they could look at the effects of different room temperatures on
the cooling process. Also, they could look at the effects of the container
on the cooling process. One important point to emphasize, is that they need
to take the temperatures in a consistent manner with respect to other constraints
(i.e., leaving the water in the pot or pouring the water into a heated cup,
if the thermometer used has an appropriate scale, etc.). Ask the students
what effects they are interested in investigating. With the use of the spreadsheet,
there seem to be an endless array of interesting things to explore!